What is Syncrystallization? States of the pH Indicator Methyl Red in Crystals of Phthalic Acid | Journal of the American Chemical Society
![SOLVED: Calculate the Ka and pKa of methyl red from the following equations and the data in the tables above; record these values in the table below (show your calculations). [H ] = SOLVED: Calculate the Ka and pKa of methyl red from the following equations and the data in the tables above; record these values in the table below (show your calculations). [H ] =](https://cdn.numerade.com/ask_images/a0a70d476fd049fea11245c72f687323.jpg)
SOLVED: Calculate the Ka and pKa of methyl red from the following equations and the data in the tables above; record these values in the table below (show your calculations). [H ] =

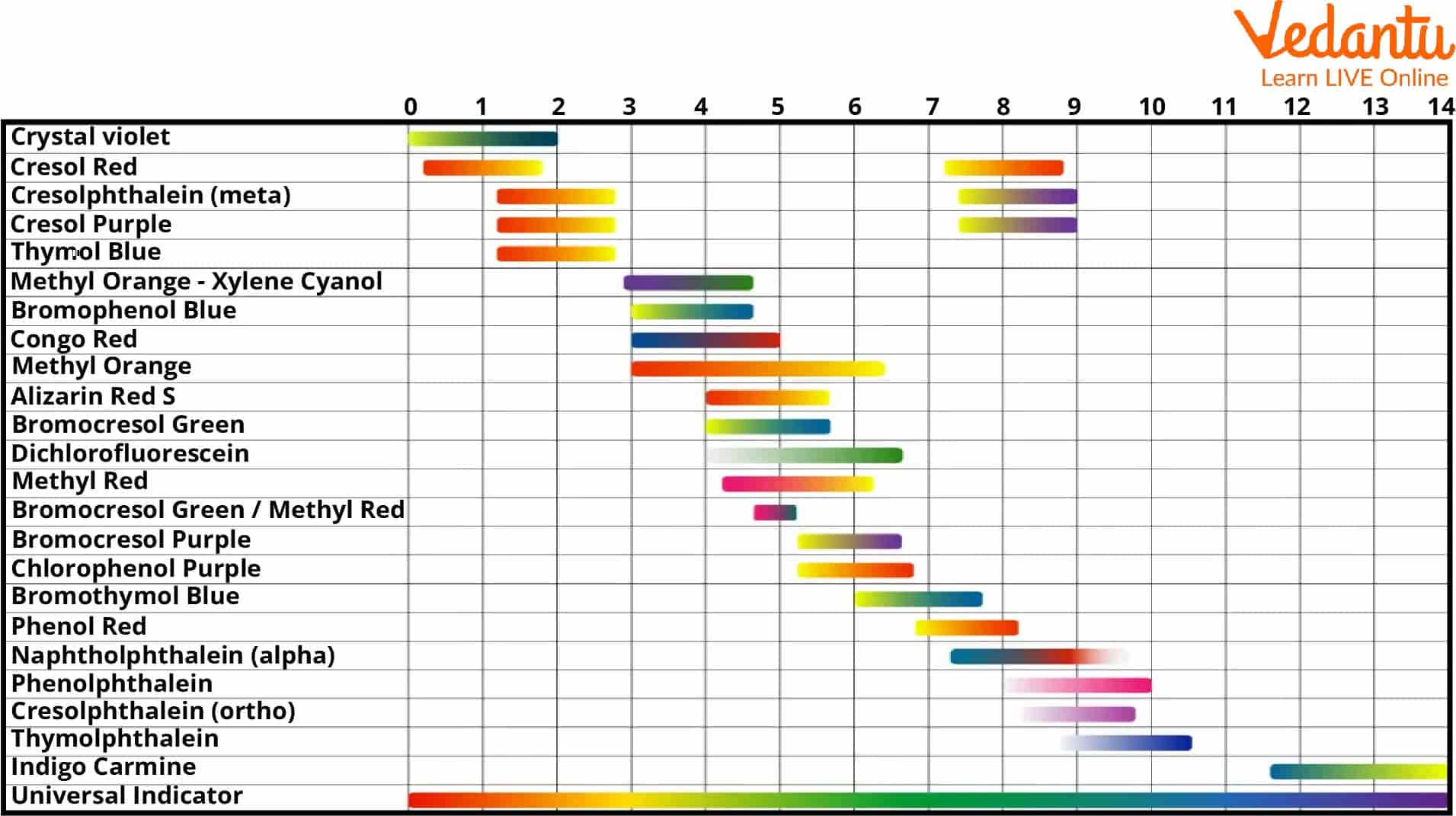
4.1 – Describe how acids and bases in solution affect certain indicators 4.2 Determine the pH of a solution using a universal indicator PH SCALE. - ppt download

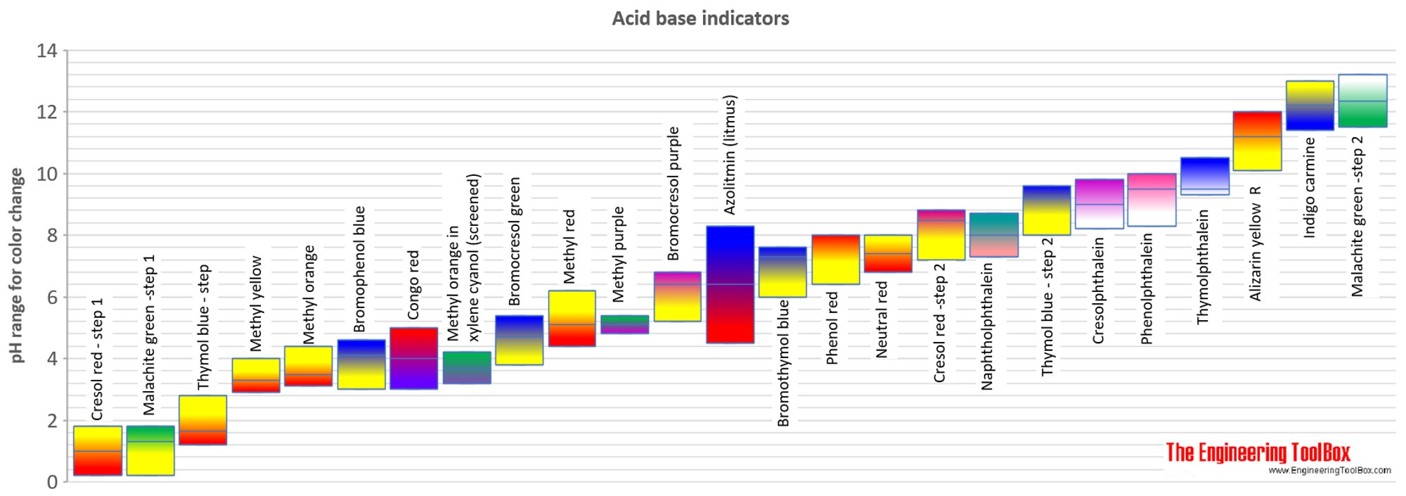
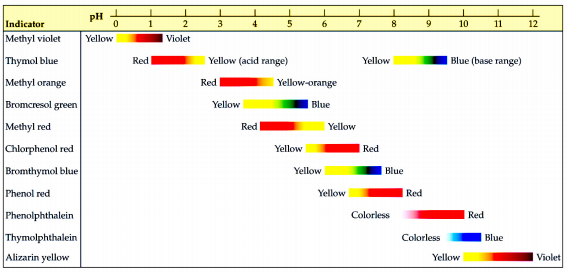
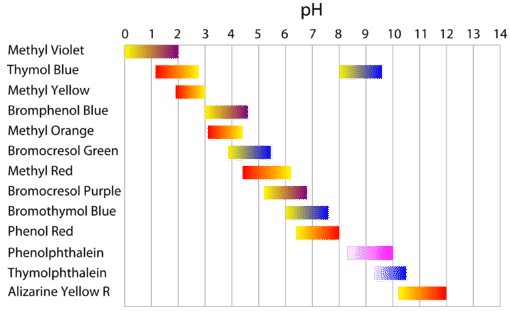
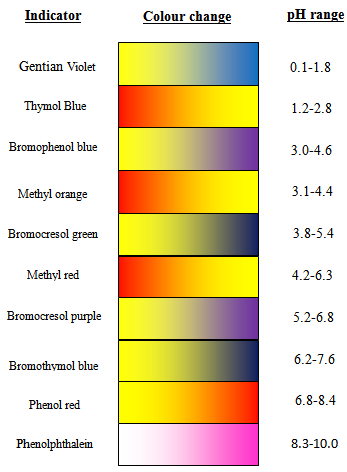
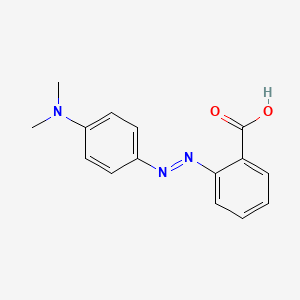
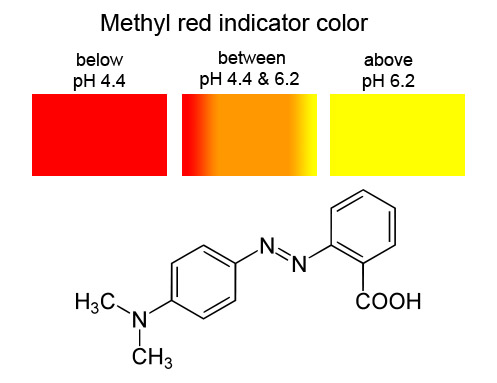
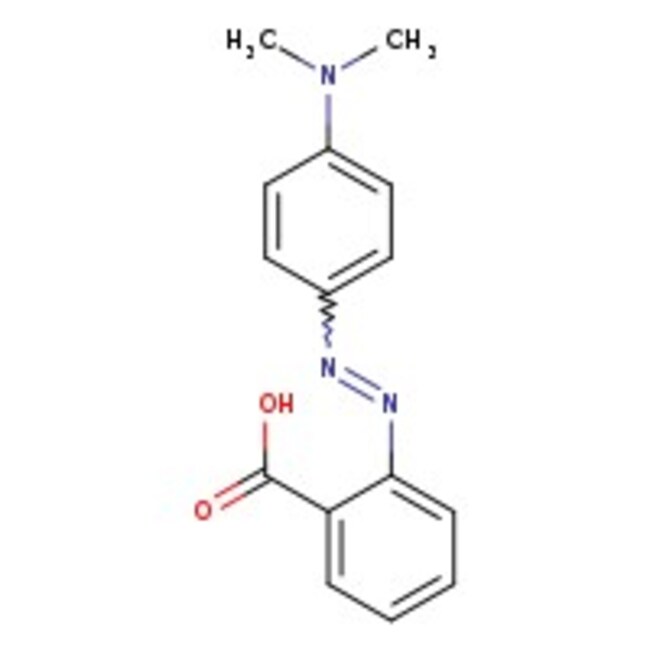
Methyl red has the following structure: It undergoes a color change from red to yellow as a solution gets more basic. Calculate an approximate pH range for which methyl red is useful.